Thursday, September 29, 2022 | 1:00 – 2:00pm ET


Thursday, September 29, 2022 | 1:00 – 2:00pm ET

September 29 – October 1


from Jul 16, 2022

Nancy Collins, PhD, RDN, LD, NWCC, FAND

In this video, David Armstrong, PhD, DPM, discusses how DHACM treatment led to reductions in amputations, hospital readmissions, and cost for patient with diabetic ulcers.
Contract Awarded for Products that Bring Improvement to the Health Care Industry
PITTSBURGH, Sept. 29, 2022 /PRNewswire/ – MolecuLight Corp., the leader in point-of-care fluorescence imaging for the real-time detection of bacteria in wounds, announces it has been awarded a new group purchasing agreement with AllSpire Health GPO, a Mid-Atlantic GPO and a partner of HealthTrust Purchasing Group, engaged with over fifty hospitals in Maryland, New Jersey and Pennsylvania. AllSpire helps health systems optimize their operations by aggregating purchasing volumes, expenses, streamlining supplier negotiations and implementing efficiencies across the supply chain. The MolecuLight i:X® and DX™ wound imaging devices, which will now be available to AllSpire’s members, are helping clinicians to improve the state of wound care and ultimately to improve outcomes.
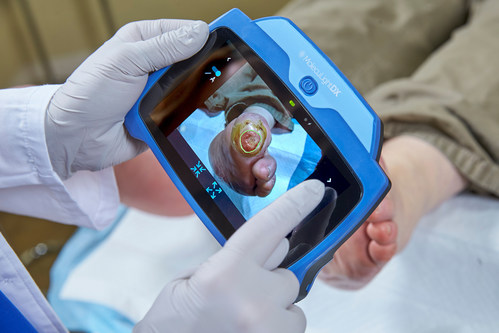
MolecuLightDX point-of-care imaging system for detection of elevated bacterial loads in wounds and for performing digital wound measurement and tracking (CNW Group/MolecuLight)
The MolecuLight imaging devices are the only FDA-approved devices that allow clinicians to visualize the presence, location, and load of bacteria (>104 CFU/g) in wounds in real-time. Published results from a recent 350-patient, 14-site clinical trial showed that the clinical standard of care alone detected 15% of wounds with elevated bacterial burden, while the addition of the MolecuLight device led to a 400% improvement in detecting these wounds2. The presence of elevated bacterial loads is known to impede wound healing1 and removal of bioburden is critical to improved wound outcomes1. The i:X and DX provide invaluable bacterial information at the point-of-care to inform clinical decision-making and enable targeted wound therapies. In a 2022 randomized controlled trial (RCT) 3, the highest level of evidence-based research, the improvement in healing rate at 12 weeks doubled in the patients that had care informed by MolecuLight fluorescence imaging compared to those that were not. Improvements in the patients’ quality of life were also reported. Another recent study reporting increased wound healing rates with the incorporation of bacterial information from MolecuLight imaging also found substantially decreased use of antimicrobial dressings and systemic antibiotics4. The MolecuLight devices also perform accurate digital wound measurement, allowing for the consistent monitoring and documentation of wounds.
“We are thrilled to have entered into a supply contract with AllSpire Health GPO,” says Anil Amlani, MolecuLight’s CEO. “Through the i:X and DX, we hope to enable significant cost-savings and improvements in clinical outcomes. AllSpire’s extensive member base can now easily access the MolecuLight wound imaging devices and see the clinical benefits in their wound care practices.”
“We are most impressed with the clinical utility that the MolecuLight i:X and DX devices provide to wound care professionals and are pleased to offer the MolecuLight portfolio via our Group Purchasing Agreement to our member hospitals”, says James Wallick, Senior Director, Strategic Sourcing at AllSpire Health GPO. “AllSpire is dedicated to sourcing the most innovative products that help to improve clinical decision-making and cost-efficiencies. We believe that the MolecuLight devices are highly innovative and will help to provide such clinical and operational benefits”.
In addition to the clinical benefits, MolecuLight procedures performed in the United States can benefit from an available reimbursement pathway including two CPT® codes for physician work to perform “fluorescence imaging for bacterial presence, location, and load” procedures and facility payment for Hospital Outpatient Department (HOPD) and Ambulatory Surgical Center (ASC) settings through an Ambulatory Payment Classification (APC) assignment.
About MolecuLight Corp.
MolecuLight Corp. is the US subsidiary of MolecuLight Inc., a privately-owned medical imaging company that has developed and is commercializing its proprietary fluorescent imaging platform technology in multiple clinical markets. MolecuLight’s suite of commercially released devices, including the MolecuLight i:X® and DX™ fluorescence imaging systems and their accessories, provide point-of-care handheld imaging devices for the global wound care market for the real-time detection of wounds containing elevated bacterial burden (when used with clinical signs and symptoms) and for digital wound measurement. The company is also commercializing its unique fluorescence imaging platform technology for other markets with globally relevant unmet needs including food safety, consumer cosmetics and other key industrial markets.
About AllSpire Health GPO
Founded in 2016, AllSpire Health GPO, LLC is a collaborative, regional group purchasing organization utilizing innovation analytics, as well as product and purchased service standardization, as a platform to escalate the improvement of clinical outcomes, enable greater access to affordable healthcare, ensure economic sustainability, and enhance patient, physician, and clinician satisfaction among its members. AllSpire Health GPO delivers value via the development and execution of clinical and service line improvement initiatives across the care continuum.
SOURCE MolecuLight



of chronic diabetic foot ulcers at the world’s premier diabetic foot conference (DFCon) in Los Angeles


Robyn Rayner, Keryln Carville, Joanna Smith and Cate Maguire

Jonathan Quinn, William Fairbairn, Gayle Silveira and Simon Platt


ALLOGRAFT APPLICATION IN ADVANCED VENOUS LEG UCLERS







Physician Fee Schedule: Changes Could Increase Amputations and Infection for Patients with Wounds, Restrict Access to Care, Impact Reimbursement



Jun Ho Park & Ji-Ung Park



Bacterial biofilms pose a therapeutic challenge to managing chronic wounds and contribute to antimicrobial resistance. Here, Powell et al. investigated the structure/activity relationships of epoxy-tigliane compounds derived from the blushwood tree with respect to their role in wound healing.

The biofilm community has historically been very successful in aggregating scientists from very diverse fields. Now, we must harness innovative technologies across disciplines to illuminate the biofilm microenvironment and create in vitro models that accurately recapitulate natural environments.

Standardized methodology demonstrates high presence of placental membrane growth factors in BioStem’sVendaje® and Vendaje AC®tissue allografts

Skeleton buried in Borneo cave suggests early artists were also early surgeons
CellResearch Corporation, a Singapore-based biopharmaceutical company today announced it has successfully closed the first Phase I study in CorLiCyte®, a stem cell therapy derived from umbilical cord lining stem cells, with research partners at the University of Colorado, Anschutz Medical Campus and ClinImmune Cell and Gene Therapy.


by Bari Weiss


Advocacy on Behalf of Members and Collaboration with Government Agencies




Guoxin Tan, Lijie Wang, Weisan Pan, Kai Chen

as First Aid Kits are Restocked During National Preparedness Month

CANTON, Mass., Sept. 07, 2022 (GLOBE NEWSWIRE) — Organogenesis Holdings Inc. (Nasdaq: ORGO), a leading regenerative medicine company focused on the development, manufacture, and commercialization of product solutions for the Advanced Wound Care and Surgical and Sports Medicine markets, today announced that company management will participate in the Morgan Stanley 20th Annual Global Healthcare Conference, which is being held at the Sheraton New York Times Square Hotel in New York, NY from September 12th-14th. Management will host a fireside chat on Tuesday, September 13th at 11:45 a.m. Eastern Time.
A live audio webcast of the conference presentation will be accessible by visiting the “Upcoming Events” section on the “Investor Relations” page of Organogenesis’s website www.organogenesis.com. An archive of the webcast will be available for replay following the conference for approximately 30 days.
About Organogenesis Holdings Inc.
Organogenesis Holdings Inc. is a leading regenerative medicine company offering a portfolio of bioactive and acellular biomaterials products in advanced wound care and surgical biologics, including orthopedics. Organogenesis’s comprehensive portfolio is designed to treat a variety of patients with repair and regenerative needs. For more information, visit www.organogenesis.com.




Lihong Chen, Yunyi Gao, Yan Li, Chun Wang, Dawei Chen, Yun Gao, Xingwu Ran
The regenerative biotechnology is reducing healing times to as little as three weeks in Mohs-related surgical wounds
PHOENIX–(BUSINESS WIRE)–Dermistat™, one of the latest wound healing innovations by BioLab Sciences, a regenerative biotechnology company, has emerged as the go-to wound healing option for dermatologists throughout the country. Introduced earlier this year, Dermistat™ is a revolutionary regenerative therapy that is transforming the wound care industry by decreasing healing time significantly compared to the standard of care secondary closure.
 Part of the MyOwn Skin™ suite of products, Dermistat™ uses a small sample of the patient’s healthy skin to create a gel-like substance that can create an autologous skin graft in as little as 48 hours. Used for acute surgical wounds, including Mohs procedures unable to be closed with standard plastic surgery techniques, and non-healing wounds, Dermistat™ is non-invasive, improves recovery time, is relatively painless, and creates only a small, superficial donor site wound.
Part of the MyOwn Skin™ suite of products, Dermistat™ uses a small sample of the patient’s healthy skin to create a gel-like substance that can create an autologous skin graft in as little as 48 hours. Used for acute surgical wounds, including Mohs procedures unable to be closed with standard plastic surgery techniques, and non-healing wounds, Dermistat™ is non-invasive, improves recovery time, is relatively painless, and creates only a small, superficial donor site wound.
“Dermistat™ was originally developed to treat severe burns, trauma and plastic procedures, however, BioLab Sciences modified the process enabling it to be effectively leveraged in the dermatologist’s office, allowing patients to recover faster and more completely,” said Jaime Leija, Chief Commercialization Officer at BioLab Sciences. “Dermistat™ is proving to be a valuable resource for Mohs patients as it eliminates the need to perform a graft to cover larger wounds and it significantly reduces the healing time.”
This novel biotechnology is based on the MyOwn Skin™ technology, which leverages a very small sample of a patient’s own skin through a non-surgical procedure to reproduce the gel-like substance within 48 hours. In many cases, Dermistat™ accelerates the healing of post-surgical and chronic wounds and because of its favorable outcomes, it is on track to disrupt the wound care industry.
“We have been using Dermistat™ on surgical defects post-Mohs surgery after excising skin cancer,” said Travis Gilbert, PA-C with Desert Dermatology & Skin Cancer Specialists. “The wounds were typically open for 5-7 days prior to applying Dermistat™. Prior to using Dermistat™, we would close with flaps mostly. Occasionally, we would use skin grafts. Today we are using mostly Dermistat™ to cover these acute surgical wounds. They are healing much more rapidly than secondary closure and do not require significant plastics closure.”
About BioLab Sciences
BioLab Sciences is a regenerative medicine company focused on creating new ways to heal the body. The company is uncovering better ways to address orthopedic injuries, wound care, pain management, aesthetic medicine, respiratory ailments, cardiovascular indications, ophthalmic issues, and more. Learn more at www.biolabsciences.net/.
Contacts
Beth Cochran, Wired PR
beth@wiredprgroup.com
(602) 758-0750

Pragateshnu Das, Debmalya Bhattacharya, Rajlaxmi Sathpathy
Mary R. Brennan MBA, RN, CWON

By Heidi Cross, MSN, RN, FNP-BC, CWON

Alton R. Johnson, Jr., DPM, FACPM, CWSP

Mandalyn Mills Luke Counterman Shanna Williams

Sara Yumeen Mélissa Roy Fatima N. Mirza Sarah Rehou Shahriar Shahrokhi


Asad Ullah, Syed I. Jawaid, Pir Naveed Ahmed Ahsan Qureshi, Tehreem Siddiqui, Khadija Nasim, Kantash Kumar, Shafqat Ullah, Mustafa Sajjad Cheema, Nikita Kumari, Hafiza Azad Elias
BEVERLY HILLS, Calif., Aug. 23, 2022 (GLOBE NEWSWIRE) — Miracle Dressing Wound Care System 21-Day Stay-in-Place Dressing has been named Wound Care Product of the Week by WoundSource, “the world’s definitive source for wound care and product information.”
Miracle Dressing™ Wound Care System is the only 21-day stay-in-place dressing. Wound prevention, monitoring, cleaning and topical applications can all be completed without dressing removal. This saves nursing time and reduces patient distress from frequent dressing changes.
The system includes Natural Marine Extract™, the ingredients of which are known to promote faster healing of the skin and better wound healing results.
The Wound Healing Society spotlighted the importance of dressings that can stay in place for an extended time, especially due to extended stays caused by COVID mitigation. They recommend utilizing dressings that decrease the intensity of wound care by avoiding the use of dressings that must be changed daily. This would allow over-stressed health care staff to reach more patients in an adequate time frame.
The Miracle Dressing System can be used for:
MBET is recognized for their skin revitalization and wound care products as well as kelp reforestation and other international environmental projects.
MBET Health is a solutions-oriented technology company focused on solving one of the most important and intransient problems facing the world of medicine: preventing and healing wounds.
MBET Health was founded by Dr. Eric Lewis, a practicing dermatologist, surgeon and scientific researcher based in Beverly Hills, CA. The MBET Health management team includes medical and surgical doctors from a multitude of disciplines, pharmacists and marine biologists.
Beginning in 2001, their founders researched a multitude of marine compounds reported to have human health benefits. As a result of their extensive studies and tests, several compounds and systems specifically designed for rejuvenation and effective repair of damaged skin have been patented (or patent-pending). The mechanisms of action of their system’s ingredients are designed to heal intractable wounds and strengthen weak, vulnerable skin to minimize the probability of breakdown.
The MBET Health website provides a destination for all wound care providers, nursing staff and senior management to see for themselves the convincing before and after photos and to learn details about proper product application and dressing maintenance.
Contact
Linda Sherman Gordon
MBET Health Chief Marketing Officer
310-243-6305
Email contact
MBET Health LinkedIn
Before / After press photos available upon request
This article was originally published here

Neal M. Lonky, MD, MPH, Jeffrey Lehrman, DPM, FASPS, MAPWCA, CPC, Andrew Schneider

