A Randomized Controlled Trial

A Randomized Controlled Trial

Two leading wound industry solutions companies, with a shared passion for improving patient outcomes, are collaborating to improve the way that Home Health and other in-home providers care for patients with chronic wounds.
SAVANNAH, GA. (PRWEB) OCTOBER 24, 2022
HARTMANN USA and Corstrata announce their collaboration to support home health and other in-home providers in caring for wound patients at a time when the incidence of complex chronic wounds is increasing while access to wound care nurse specialists is becoming more challenging. At the core of a successful wound care program is access to both highly effective advanced wound dressings as well as clinical expertise to implement evidence-based treatment protocols and monitor wounds to closure. According to a recent study published by top-50 accounting firm BerryDunn, National Healthcare at Home Best Practices and Future Insights Study, 100% of Home Health Centers of Excellence (those Home Health agencies in the top 10% for quality and patient satisfaction and with a positive financial surplus) have a wound-certified specialist on staff.
This collaboration will increase access to Corstrata’s team of virtual board-certified wound nurses (WOC nurses) and HARTMANN’s suite of advanced wound care products to improve clinical and financial outcomes for in-home providers that care for patients with chronic wounds.
HARTMANN has been providing advanced wound dressing solutions globally for over 150 years and has evolved with its broad portfolio of high-quality, cost-effective products that provide home health clinicians with a simplified, consistent approach for effectively managing wounds. Corstrata provides virtual wound and ostomy care management nationwide across multiple provider settings, including home health, skilled nursing facilities, hospice, and emerging hospital-at-home solutions companies.
According to Jon Procopio, Managing Director of HARTMANN USA, “Patient care is our priority. HARTMANN strives to enable the progression of the wound towards complete healing that patients deserve and strengthen the confidence that healthcare professionals need to provide wound care. We have a nationwide team of dedicated account and customer care representatives specifically trained for consultation, education, and support in offering clinical and business solutions. Now, with Corstrata, we will enhance access to clinical expertise related to wound care through their team of certified WOC nurses.”
“The Corstrata team is excited about this important collaboration with HARTMANN to create access to Corstrata’s virtual WOC nurses for customers and the patients they serve. With up to one-third of all home health and hospice patients having a chronic wound, it is critical for clinicians to provide evidence-based care to both prevent and heal wounds,” says Joseph Ebberwein, co-founder and Chief Financial Officer of Corstrata. “At this time when agencies are struggling with critical staffing shortages, including WOC nurses, and increasing financial challenges, having a strong wound program is essential. This collaboration between Corstrata and HARTMANN provides a path to success.”
According to Katherine Piette, Corstrata’s CEO, the decision to collaborate with HARTMANN is an easy one. “Our virtual WOC nurses rely on our provider customers having access to highly effective advanced wound dressings to accelerate wound healing and reduce the overall cost of patient care, ” Piette says. “HARTMANN has a unique suite of advanced dressings that are being used by some of the top home health providers in the U.S. with impressive results. We are excited about the opportunity to improve the level of wound care provided for this ever-growing cohort of complex wound patients. Our clients can access clinical support from Corstrata when needed without the cost of hiring their own WOC nurse, a costly and often frustrating proposition. This collaboration will equip providers with turn-key wound solutions that they have been missing in their clinical care delivery at a crucial time in the industry.”
About HARTMANN
The HARTMANN GROUP is one of the leading providers of wound treatment and skin integrity solutions around the world. Wound dressings and maintaining healthy skin have been at the heart of HARTMANN from the beginning when we introduced the world’s first antiseptic wound dressing over 150 years ago. Overall, HARTMANN looks at rich legacy. Every day, healthcare professionals and patients rely on HARTMANN brands in the segments of Incontinence Management (e.g. MoliCare®), Wound Care (e.g. Zetuvit®) and Infection Management (e. g. Sterillium®). This is expressed in our brand promise of “Helps. Cares. Protects.” In 2021, the HARTMANN GROUP reported Group sales of EUR 2.3 billion.
For the latest information on HARTMANN, follow @HARTMANN_GROUP on Twitter.
To learn more about the HARTMANN GROUP, click here.
To learn more about HARTMANN USA, click here.
About Corstrata
Corstrata is a virtual care solution that utilizes technology to provide access to scarce certified wound and ostomy nurses at the patient’s bedside in post-acute provider settings, including home health, hospice, skilled nursing facilities, and emerging hospital-at-home providers. Corstrata’s team of WOC nurses provides consultations with provider staff at the patient’s bedside, either through HIPAA-compliant video or through review of store-and-forward wound images, to improve clinical and financial outcomes for providers.
For the latest information on Corstrata, follow @Corstrata on Twitter.
To learn more about Corstrata, click here.
This article was originally published here
From Feb 7, 2022 – David Armstrong, Desmond Bell, Rick Zollinger, Randy Cook, Rhonda Crowe

Liqiong Yuan 1, Maoting Ye, Ting Yang


Negin Shamsian, Heather Hettrick, Adriano Mehl, Tobe Madu, Mark Barakat, Windy Cole, Michael Oliver

Schaum, Kathleen D. MS

Pasek, Jarosław MD, PhD; Stanek, Agata MD, PhD; Szyluk, Karol MD, PhD; Cieślar, Grzegorz MD, PhD
major differences in immune response-modulatory effects


Fay Crawford, Donald J. Nicolson, Aparna E. Amanna & Marie Smith

A Prospective Comparative Study – Jamuna Nagaraj, Venkatesh Subbiah

Audrey Abella
Indre Jasineviciute, Juozas Grigas, Gintare Ziukaite, Arnoldas Pautienius, Dainius Razukevicius, Judita Zymantiene & Arunas Stankevicius


Wed, Nov 2, 2022 · 2:30 PM Eastern Time (US & Canada) (GMT -4:00)

ADVANCED WOUND CARE SUMMIT


Muñoz, Victoria; Pino, Ander; Martinez, Carmen; Echevarria, Begoña; Lacramioara, Varlan; Anitua, Eduardo


Neil Baker OBE

Jennifer Spector, DPM, FACFAS, Assistant Editorial Director

A Case Study Pérez-Acevedo, Gemma; Bosch-Alcaraz, Alejandro; Torra-Bou, Joan Enric

Jennifer Spector, DPM, FACFAS, Assistant Editorial Director



Sylvie Hampton

A Multiple Case Series

Dr. Abigail Chaffin
surpassing 2,000 peer-reviewed medical journal studies published
ST. PAUL, Minn., Oct. 13, 2022 /PRNewswire/ — 3M Health Care’s Medical Solutions Division today announced its 3M™ V.A.C.® Therapy negative pressure wound therapy (NPWT) has surpassed a clinical evidence milestone of 2,000 published, peer-reviewed medical journal studies. V.A.C. Therapy is the first and only NPWT solution to garner this number of published studies about its therapy. It is backed by more clinical data than any other brand, accounting for more than 75% of published NPWT clinical evidence.
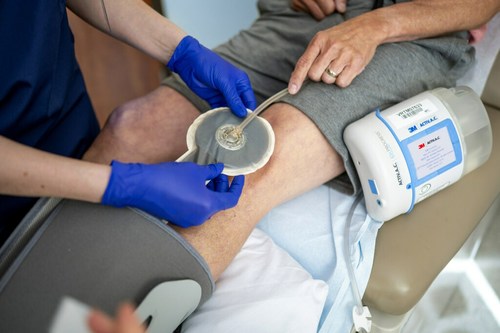
3M™ V.A.C.® Therapy negative pressure wound therapy (NPWT) has surpassed a clinical evidence milestone of 2,000 published, peer-reviewed medical journal studies. It is backed by more clinical data than any other brand, accounting for more than 75% of published NPWT clinical evidence.
The clinical studies have been conducted by wound care professionals worldwide and published in journals across the globe, covering a comprehensive range of wound types, wound care settings and study formats, such as case studies, economic studies, randomized controlled trials and more.
“Clinical evidence has always been a foundational element to establishing credibility for V.A.C. Therapy and our NPWT products in the wound care community,” said Ronald Silverman, M.D., 3M Health Care senior vice president of clinical affairs and chief medical officer. “Published studies have also helped to promote adoption of NPWT and spur therapy innovations, including 3M™ Prevena™ Therapy for incision management, 3M™ Veraflo™ Therapy for instillation therapy for open wounds, 3M™ AbThera™ Open Abdomen Negative Pressure Therapy. Our team members in the field are also actively engaged with wound care experts worldwide, working right alongside clinicians to observe the changing nature of wound care and gather feedback about our products, which helps us identify opportunities for innovation.”
Today, V.A.C. Therapy is used across a spectrum of health care settings, from acute care facilities to ambulatory surgical centers, assisted living facilities, and in patients’ homes. In the U.S., V.A.C. Therapy is available with 24/7 remote therapy monitoring to support adherence to the therapy. 3M’s NPWT portfolio continuously evolves to meet clinician and patient needs. Last year, 3M launched the first-ever silicone-acrylic hybrid drape for use with V.A.C. Therapy, the 3M™ Dermatac™ Drape, an innovation designed to be gentle on patients’ skin and easy for clinicians to use.
“Today’s wound care patients are often sicker and have more comorbidities, making their wounds more complex to treat and increasing the demands on clinicians’ time. 3M strives to provide a robust tool selection to address clinicians’ unique wound care needs and make our products easier to use to help save their valuable time — and ultimately, help transform outcomes and improve lives for wound care patients,” said Dr. Silverman.
For more information, visit www.3m.com/npwt.
About 3M
3M (NYSE: MMM) believes science helps create a brighter world for everyone. By unlocking the power of people, ideas and science to reimagine what’s possible, our global team uniquely addresses the opportunities and challenges of our customers, communities, and planet. Learn how we’re working to improve lives and make what’s next at 3M.com/news or on Twitter at @3M or @3MNews.
Photo – https://mma.prnewswire.com/media/1919999/3M_VAC_Therapy.jpg
Logo – https://mma.prnewswire.com/media/1343410/3M_Logo.jpg
SOURCE 3M
This article was originally published here


Michael King, Chief Medical Officer, Upperline Health
Studies of Wound Closure Rate with Novel Chronic Wound Treatment Continue
SARASOTA, Fla., Oct. 13, 2022 /PRNewswire/ — Omeza today announced that the Centers for Medicare and Medicaid Services (CMS) has confirmed a HCPCS reimbursement code for Omeza® Collagen Matrix; code A2014, “Omeza collagen matrix, per 100 mg” was established to describe the product.

Omeza® Collagen Matrix is the first of its kind drug-device combination product, with a simple snap and squeeze application for chronic wounds.
Omeza® Collagen Matrix is the first of its kind drug-device combination product, with a simple snap and squeeze application for chronic wounds. The FDA-cleared drug-device received a Level II Healthcare Common Procedure Coding System (HCPCS) reimbursement code paving the way for providers to receive reimbursement from Medicare.
The decision came after application was made to CMS in late 2021. Omeza Chief Commercial Santino Costanza stated, “This decision by CMS opens the doors to Omeza’s innovative treatment line for millions of Americans covered by Medicare who are currently suffering from chronic wounds. Now we look forward to educating commercial payors on the health, financial and humanitarian benefits of a positive reimbursement decision.”
Earlier this year the Department of Veterans Affairs Federal Supply Schedule (FSS) Service granted contract status for Omeza® Collagen Matrix. All Omeza products are available to government agencies through Marathon Medical, a prime vendor for the VA.
Currently, three US clinical trials are investigating the use of the three-product Omeza treatment product line, which includes Omeza® Collagen Matrix, for documentation of healing rates in venous ulcers, diabetic ulcers, and other chronic wounds. Concurrently, individual case studies submitted by providers testing the Omeza treatment product line on chronic wound closure in their clinical settings report an average percentage area reduction (PAR) of 60% at 4 weeks.
Omeza ® Collagen Matrix is indicated for the management of wounds including partial and full-thickness wounds, pressure ulcers, venous ulcers, diabetic ulcers, chronic vascular ulcers, tunneled/undermined wounds, surgical wounds (donor sites/grafts, post-Moh’s surgery, post-laser surgery, podiatric, wound dehiscence), trauma wounds (abrasions, lacerations, superficial partial thickness burns, skin tears) and draining wounds.
Omeza ® Collagen Matrix is the first drug/device combination product to deliver an anhydrous 3- dimensional microstructure of collagen to challenging wounds. When applied to a wound surface, the snap and squeeze matrix is naturally incorporated into the wound over time. Omeza® Collagen Matrix is designed for intimate contact with both regular and irregular wound beds, to provide a conducive environment for the patient’s natural wound healing process.
About Omeza:
Omeza (www.omeza.com) is a skin science company pursuing equitable access to better wound care outcomes for patients at all sites of care. The company is based in Sarasota, FL USA. Inquiries from medical and health professionals should be directed to info@omezapro.com.
SOURCE Omeza LLC
This article was originally published here


aimed at regulated health professionals in Ontario

Nancy Collins, PhD, RDN, LD, NWCC, FAND Giovanna Rosario Arroyo, MA, RDN, CD, LDN




2022 Fall Symposium on Advanced Wound Care (SAWC)

NEWS PROVIDED BY
American College of Wound Healing and Tissue Repair
Oct 11, 2022, 09:00 ET
American Society of Plastic Surgeons and American College of Wound Healing and Tissue Repair to Discuss Fellowship Training to Improve Patient Outcomes
BOSTON, Oct. 11, 2022 /PRNewswire/ — The American Society of Plastic Surgery (ASPS) and the American College of Wound Healing and Tissue Repair (ACWHTR) will hold a joint strategic planning meeting on Oct. 27 in Boston to discuss advanced fellowship training in wound healing and tissue repair.
Since 2011, ACWHTR has trained fellows in wound healing and tissue repair at the University of Illinois at Chicago and helped create similar university-based programs that focus on training non-surgeons in the field of wound healing.
“With more than 6.2 million people in the United States living with non-healing wounds – magnified by an aging society as well as epidemics of obesity and diabetes, the number of patients with these conditions continues to grow,” says ASPS President J. Peter Rubin, MD, MBA. “Over the past year, ASPS and ACWHTR have engaged in dialogue about establishing new training paradigms for this much-needed and constantly evolving clinical field.”
Non-surgical providers lead many wound care centers; however, surgeons who provide the needed surgical procedures are also increasingly taking on leadership roles as medical directors, in-patient service chiefs, and leaders in the field.
This strategic planning session is open to all interested providers, medical and surgical professional society representatives, and those currently directing surgical, non-surgical or hybrid training programs. The goal of the meeting is to improve patient outcomes across many settings by establishing a formal, consistent educational curriculum. ASPS has led the process and proposes a non-ACGME match program for plastic surgery residents to build on the trainee’s foundational knowledge in skin and wound care procedures.
“A further objective of the collaboration is to increase the number of non-surgical fellowships based on the current ACWHTR educational platform,” says president and founder of ACWHTR, William J. Ennis. DO, MBA.
The strategic planning and ASPS/ACWHTR Wound Care Fellowship launch meeting will take place at 1:15 p.m. EDT at the Boston Convention & Exhibition Center on Oct. 27, during Plastic Surgery The Meeting in Boston. The option to participate virtually is available. Kindly respond to ASPS Senior Vice President Gina T. McClure at gmcclure@plasticsurgery.org for additional information or to register for the virtual event.
About American College of Wound Healing and Tissue Repair
The American College of Wound Healing and Tissue Repair (ACWH) is a 501(c)(3) nonprofit organization based in Chicago that is committed to advancing the field of wound care through education, research, and advocacy. The College fosters the training of medical professionals through the sharing of a physician-based, clinical fellowship curriculum developed in conjunction with the University of Illinois Hospital and Health Sciences System and allied healthcare colleges and programs. The goal of the organization is to designate wound care as a board-certified medical specialty.
About the American Society of Plastic Surgeons
The American Society of Plastic Surgeons (ASPS) is the largest organization of board-certified plastic surgeons in the world. Representing more than 7,000 physician members, the society is recognized as a leading authority and information source on cosmetic and reconstructive plastic surgery. ASPS comprises more than 93 percent of all board-certified plastic surgeons in the United States. Founded in 1931, the society represents physicians certified by The American Board of Plastic Surgery or The Royal College of Physicians and Surgeons of Canada.
SOURCE American College of Wound Healing and Tissue Repair
This article was originally published here
FREDERICK, MD. (PRWEB) OCTOBER 11, 2022
Nearly 6 million people in the U.S. suffer with chronic wounds. That care translates to more than $96.8 billion in annual Medicare costs. Nanobiofab, a nanotechnology startup founded by Dr. Xiaonao Liu, is working to make detection and care of such wounds quicker and more efficient by utilizing their proprietary real-time agnostic patch for infection detection, an intelligent nose known as RAPID-iNose.
The company is a member of the Frederick Innovative Technology Center, Inc. and was recently awarded a $250,000 Defense Health Agency SBIR Program contract for their proposal, “RAPID-iNose: Real-time Agnostic Patch for Infection Detection using Intelligent Nose.” The project combines a patented, wireless and highly-sensitive nanosensor array with artificial intelligence algorithms. In tests to date, the RAPID-iNose automatically and continuously captures information on the types and amounts of pathogens. Results can then prompt infection alerts to patients and doctors, eliminating some of the biggest challenges in wound care.
Dr. Liu explains, “Currently, clinical judgment is required for diagnosis and treatment. However, symptoms of wound infection are commonly masked in patients with complex wounds.”
Considered the current “gold standard,” culture-based antimicrobial susceptibility testing is now conducted under aseptic conditions in laboratories. This requires assessment by specialists using reagents, which may not be logistically feasible, especially in a battlefield environment. Turnaround is also slow, taking anywhere from hours to days for processing, depending upon transportation of the sample. Caregivers lack the means for continuous sampling to monitor patient progress. And there is no Food and Drug Administration approved, telemedicine-capable, deployable device for early detection of wound infections or real-time monitoring in either field environments or hospital settings.
Nanobiofab’s previous work with the Small Business Innovation Research Program includes efforts to improve medical training through AI-enhanced clinical simulators with West Virginia University’s Center for Simulation Training and Education for Patient Safety. Kathie Callahan Brady, Nanobiofab advisor and FITCI CEO, says, “This company creates revolutionary million-scale nanomaterial that can be used in wide-ranging applications, from routine health monitoring to cancer detection, and everyone is an opportunity to improve the lives and reduce suffering.”
RAPID-iNose will be the world’s first wearable intelligent device for real-time monitoring of wound infection, replacing current resource-intensive detection methods. The device has the potential to help clinicians identify pathogens quickly, safely and easily, allowing for effective use of antimicrobials as well as reducing treatment costs substantially.
For more information on Nanobiofab or their nanosensor research, log on to http://www.nanobiofab.com.
This material is based upon work funded by the Defense Health Agency Small Business Innovation Research /Small Business Technology Transfer Programs under US Army Medical Research Acquisition Activity.
Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the Department of Defense, DHA SBIR/STTR Programs or USAMRAA.
This article was originally published here


Technique in the Closure of Open Fracture Wound and Infected Wound With Significant Skin Loss

Alisha Oropallo, MD, FACS, FSVS, FAPWCA, FABWMS







and Ostomy® announces partnership to award wound care scholarships in Barbados




Dressing Demonstrates Bioabsorption and Biocompatability in Internal Bleeding


Readers who regularly frequent these pages are probably familiar with the topic of wound care.
One of the common causes of amputation of lower extremities


Tuesday October 11th, 12:00PM – 1:00PM EDT

BY PAUL KESSELMAN, DPM

By Dr. Lenz and Dr. Hussain

Amelia Beatty, DNP, MSN, NP, RN, CWCN-AP Penny S. Jones, AGNP-C, MN, CWS, CWCN-AP, COCN Christopher Vail, PA-C, MMCi Julie A. Thompson, PhD Staci S. Reynolds, PhD, RN, ACNS-BC, CCRN, CNRN, SCRN, CPHQ

Terry Treadwell

A Clinical Experience in Two Patients Nima Khavanin and Hooman T. Soltanian



Christine Miller, DPM, PhD

Laura Swoboda, DNP, APRN, FNP-C, FNP-BC, CWOCN-AP

by Caroline Fife, M.D.



Lower Extremity Wounds in Patients With Diabetes: A Case Series

Therapy System after Evaluation Yields Positive Results


