
Month: August 2022


Chitosan/Sodium Alginate/Velvet Antler Blood Peptides Hydrogel Promotes Diabetic Wound Healing
via Regulating Angiogenesis, Inflammatory Response and Skin Flora


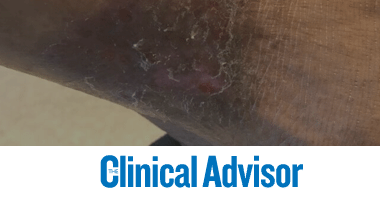
Lower Leg Ulcers
Briana Fernandez, Sidra Deen, Carly Dunn, Tara L. Braun



Application of an instructive hydrogel accelerates re-epithelialization of xenografted human skin wounds
Holly D. Sparks, Serena Mandla, Katrina Vizely, Nicole Rosin, Milica Radisic & Jeff Biernaskie


A Retrospective Cohort Study on Diabetic Foot Disease: Ascertainment of Ulcer Locations by Age Group
Patrícia Rosinha, Miguel Saraiva, Lia Ferreira, Susana Garrido, André Carvalho, Cláudia Freitas, Cláudia Amaral, Luís Costa, Luís Loureiro, Rui Carvalho



The Evolution of Platelet-Based Wound Therapy
Shivani J. Patel, BS Khurram Khan, DPM, FACFAS, FACPM James McGuire, PT, DPM, CPed, FAPWHc

Best Wound Care Therapy I Have Ever Experienced
Posted by Tim Gilmer in Life After Paralysis on August 12, 2022

Wound Expo 2022
MANCHESTER CENTRAL, 22ND SEPTEMBER 2022
BioDerm, Inc. Readies for Direct-to-Consumer Growth with Executive Additions
LARGO, Fla.–(BUSINESS WIRE)–BioDerm, Inc., a leading provider of disposable medical devices and wound care supplies, announced the addition of two executives to strengthen its leadership position in providing premium solutions for patients with chronic medical conditions. BioDerm President & CEO Gaet Tyranski said, “I’m ecstatic to add seasoned talent in the areas of sales and marketing and compliance to take us to the next level.”
 Colleen Kennedy is joining BioDerm as Vice President of D2C Sales and Marketing, bringing more than 20 years’ experience driving D2C marketing programs. Most recently, Colleen directed agency teams at Bluewater Media, working with a variety of D2C brands to maximize revenue opportunities. She specialized in multi-channel D2C programs that combined both traditional and emerging media channels to generate positive results through data driven analysis. Colleen has the unique ability to talk strategy in lay terms to guide teams in new and profitable directions, while building relationships based on trust and confidence. Prior to Bluewater Media, Colleen was the VP Account Director at D2C agency Acquirgy.
Colleen Kennedy is joining BioDerm as Vice President of D2C Sales and Marketing, bringing more than 20 years’ experience driving D2C marketing programs. Most recently, Colleen directed agency teams at Bluewater Media, working with a variety of D2C brands to maximize revenue opportunities. She specialized in multi-channel D2C programs that combined both traditional and emerging media channels to generate positive results through data driven analysis. Colleen has the unique ability to talk strategy in lay terms to guide teams in new and profitable directions, while building relationships based on trust and confidence. Prior to Bluewater Media, Colleen was the VP Account Director at D2C agency Acquirgy.
BioDerm promoted Alicia Lance to Sr. Director of Compliance for BioDerm and all affiliates. Alicia started at BioDerm in 2013, as a Manager of AMDI and most recently served as the Director of Customer Care and AMDI. She has built a strong customer care program at BioDerm based on integrity and dedication to patient outcomes. Alicia serves on the Medical Supply committee for AAHomecare and has over 20 years’ experience working in DME operations and compliance. She is excited to direct and expand BioDerm’s compliance program to enhance the company’s growth. Prior to BioDerm, Alicia worked at CCS Medical as a Compliance Manager. She holds a BS in Allied Health from the University of West Alabama.
About BioDerm
Headquartered in Largo, Florida, BioDerm is a leading provider of disposable medical devices and wound care supplies to patients with chronic conditions. BioDerm manufactures proprietary hydrocolloid products for urinary management, securement, infection control and skin protection, challenging accepted inferior standards of care by creating products that reduce infection rates, add comfort and reliability, and vastly improve quality of life. BioDerm’s flagship products for male urinary incontinence are Men’s Liberty for the home setting and Men’s Liberty Acute for in-patient settings. Other products include CathGrip for securement and BioPlus and FreeDerm for skin protection.
Wound Care Resources (“WCR”), a subsidiary of BioDerm, provides infection control products and other wound management supplies to patients with ventricular assist devices (VADs) installed due to heart failure, as well as other conditions.
For more information on BioDerm’s line of products visit www.bioderminc.com.
Contacts
Amy Stephens
Director of Marketing
727-416-2684
View source version on businesswire.com: https://www.businesswire.com/news/home/20220817005217/en/

Buckmaster Show: The new spike in the Diabetes Epidemic
From 6/25/2019
Untreated chronic wounds cost Australia billions, medical groups say | ABC News
Medical groups say untreated chronic wounds are costing the Australian health systems $3 billion a year. Subscribe: http://ab.co/1svxLVE
Around Australia there are 420,000 people living with chronic wounds, with those living outside the big cities, poorer people and First nations people the worst affected.
Hayley Ryan, chair of advocacy group Wounds Australia, speaks with ABC News.


The Pros and Cons of Wound Care Nursing
Natalie Vaughn

How to Care for Periwound Skin
Periwound skin, found around a wound, is fragile and prone to injury

Squamous Cell Carcinoma of the Scalp
David D Zabel, MD, FACS
They wanted to amputate below the knee. For now my treatment is going better in Turkey.
It is not clear what methods Dr Yusuf Maraşlı uses to reduce amputations, this might be worth a watch anyway:
Diabetic Limb Salvage conference joins 2023 Symposium on Advanced Wound Care (SAWC) Spring
Wound Healing Society for immersive educational experience
Premier annual limb salvage meeting co-locates with the leading meeting dedicated to the research, management, treatment, and prevention of wounds in April.
Three industry-leading wound care organizations are uniting for a conference in April 2023, focusing on wound prevention and management, research, and limb salvage. The meeting will offer the most cost-effective, comprehensive, and immersive educational event for professionals in the wound care space.
The Symposium on Advanced Wound Care (SAWC) Spring | Wound Healing Society (WHS) is the leading meeting dedicated to the research, management, treatment, and prevention of wounds; and Diabetic Limb Salvage (DLS) is the premier annual limb salvage education event focused on wound healing and preventing amputations. The 2023 event is organized by HMP Global, the omnichannel leader in healthcare events and education, and will be held April 26-30, 2023, in National Harbor, Maryland.
The in-person event will provide opportunities for networking and collaboration, with an educational agenda featuring an expert lineup of faculty, in-depth discussions, and exposure to innovation, proven techniques, and effective strategies for patient care. The interdisciplinary agenda is designed for every clinician interested in wound care, including physicians, nursing professionals, physical therapists, researchers, scientists, podiatrists, and dietitians — connecting the entire wound care team with the foremost experts in the field to improve patient outcomes through education.
DLS Co-Chairs are Dr. Christopher E. Attinger, Chief, Division of Wound Healing at MedStar Georgetown University Hospital and professor of plastic and orthopaedic surgery, Georgetown University School of Medicine in Washington, D.C.; and Dr. John S. Steinberg, Co-Director, MedStar Health Wound Healing Institute at MedStar Georgetown University Hospital, Hospital Center Director of the Podiatric Residency Training Program, MedStar Health, and a professor of plastic surgery at Georgetown University School of Medicine.
“Through this collaboration, educational opportunities at the conference will focus on every aspect of wound research, prevention, and healing along with a focus on limb salvage,” said Dr. Steinberg. “We are solving the issue of access to education by uniting these three events this year to provide learners with one comprehensive event. It is an opportunity for providers to strengthen their clinical skills, invigorate their approach, and positively impact their ability to care for their patients.”
The collaboration with DLS will add more limb salvage-focused topics to the conference agenda, including:
- Advanced Surgical Options for Lower Extremity Limb Salvage
- Diagnosing and Managing the Charcot Foot
- Integration of Plastic Surgery with the Limb Salvage Team
- Endovascular Intervention
The WHS President is Dr. Kenneth Liechty, Division Chief of Pediatric Surgery and Director of Fetal Medicine, University of Arizona, and Surgeon in Chief of Diamond Children’s Hospital; and Co-Chairs are Dr. Daria Narmoneva, associate professor, University of Cincinnati, and Dr. Carlos Zgheib, assistant professor of surgery, University of Colorado Denver School of Medicine in Aurora.
“Although each of these three organizations have a unique mission, we are united in our goals of improving outcomes for patients and patient populations,” Dr. Liechty said. “We are excited to host one symposium for every member of the wound care team, allowing us to provide the highest caliber training and education that all clinicians can incorporate in their practice.”
SAWC Spring Co-Chairs are Dr. Robert S. Kirsner, Harvey Blank Professor and chairman, Dr. Philip Frost Department of Dermatology and Cutaneous Surgery, University of Miami Miller School of Medicine; and Dot Weir, RN, CWON, CWS, clinician at Saratoga Hospital Wound Healing.
“We have partnered with the Wound Healing Society for 15 years, providing a robust educational experience for meeting participants, and this year’s event will be even stronger with the addition of multiple topics on amputation prevention,” Dr. Kirsner said. “No other wound care conference offers the level of education, advanced state-of-the-art clinical reviews, and emerging research findings.”
For more information or to register, visit sawcspring.com.
ABOUT HMP GLOBAL
HMP Global is the force behind Healthcare Made Practical — and is an omnichannel leader in healthcare content, events, and education, with a mission to improve patient care. The company produces accredited medical education events — in person and online via its proprietary VRTX virtual platform — and clinically relevant, evidence-based content for the global healthcare community across a range of therapeutic areas. Its brands include the HMP Global Learning Network, healthcare’s most comprehensive source for news and information; Psych Congress, the largest independent mental health meeting in the U.S.; the Evolution of Psychotherapy, the world’s largest independent educational event for mental health professionals; the Leipzig Interventional Course (LINC), the leading, global gathering for interdisciplinary cardiovascular specialists; EMS World Expo, North America’s largest EMT and paramedic event; and the Symposium on Advanced Wound Care (SAWC), the largest wound care meeting in the world. For more information, visit hmpglobal.com.

Lymphedema – Assessment and Management FAQ
Alex K. Wong MD, FACS,

Abscess of the Left Foot: Amputation Sites Treated With Placental and Umbilical Tissue
Jason Anderson, DPM, FACFAS

Postoperative Infection in an Immunocompromised Patient
Charles B Parks, DPM, FACFAS
Regenative Labs announces groundbreaking Wharton’s Jelly research demonstrating HCT/P compliance after processing
This includes the analogous nature of articular cartilage, muscle fascia, and intervertebral disc confirmed by way of comparative Scanning Electron Microscope analysis
PENSACOLA, Fla., Aug. 15, 2022 /PRNewswire/ — Regenative Labs, a leading HCT/P manufacturer, has co-authored a pioneering paper together with experts from The Institute of Regenative Medicine and the Department of Pharmacology and Chemical Biology, Baylor College of Medicine.
“This paper is a market disruptor and will be our most significant paper released to date. This is the first literature that we are aware of to utilize Scanning Electron Microscope (SEM) images of actual tissue samples to objectively demonstrate on a qualitative and quantitative basis that collagen structural tissue matrices in our post-processed Wharton’s Jelly allografts and those in articular cartilage, muscle fascia, and intervertebral discs are analogous,” said Regenative Labs CEO, Tyler Barrett.
This research highlights our commitment to the Regenerative Medicine community. We believe the combination of our IRB-approved observational studies, peer-reviewed publications, ISO-certified laboratory processes, and our commitment to compliance with FDA and American Association of Tissue Banks (AATB) standards, sets the standard for HCT/P manufacturers. Regenative Labs has pioneered the use of perinatal tissue allografts and is pleased that this paper supports our current homologous use practices, consistent with our 361 status.
Currently, the treatments for the Intervertebral Disc (DDD) range in cost and effectiveness from an $8 bottle of Ibuprofen to $150,000 for spinal fusion (1). Neither of these treatment options target the foundational issue of ECM cartilage breakdown in the intervertebral discs. By age 35, 30% of people show signs of DDD; by age 60, this increases to 90% (2). That we are aware of, this is the first perinatal tissue allograft in the medical marketplace that may be applied in a homologous fashion per FDA 361 guidelines to replace or supplement missing or damaged connective tissue. All other non-surgical paradigms focus on symptom management and do not address the disc’s collagen structural degeneration. In collaboration with medical providers across the country, we are actively investigating additional homologous use applications for this technology in tissue defects associated with the load-bearing joints of the knee, hip, shoulder, spine, ankle, and foot.
Billions of dollars are spent annually on the surgical care and treatment of patients suffering from degeneration of load-bearing joints and intravertebral discs. We are honored to offer patients evidence-based and structural tissue defect-specific non-surgical applications on a global scale.
Additional Sources:
- How much does degenerative disc treatment cost? CostHelper. (n.d.). Retrieved August 10, 2022, from https://health.costhelper.com/degenerative-disc.html
- Degenerative disc disease. Columbia Neurosurgery in New York City. (2021, July 21). Retrieved August 10, 2022, from https://www.neurosurgery.columbia.edu/patient-care/conditions/degenerative-disc-disease
About Regenative Labs: Regenative Labs produces regenerative medicine products to address the root cause of a patient’s conditions using Wharton’s Jelly innovations rather than masking the pain with other treatments. Regenative Labs works closely with scientists, physicians, hospitals, and surgery centers to constantly monitor and improve patient progress and outcomes for new product development. Formed by veteran industry professionals familiar with daily challenges of innovations in healthcare, the company provides non-addictive, non-invasive options for patients. Regenative Labs’s expert product research and development team complies FDA guidelines of minimal manipulation for homologous use. The company adheres to AATB and FDA guidelines. Learn more at Regenative’s website: www.regenativelabs.com
SOURCE Regenative Labs

Metagenomic features of bioburden serve as outcome indicators in combat extremity wounds
Aram Avila-Herrera, James B. Thissen, Nisha Mulakken, Seth A. Schobel, Michael D. Morrison, Xiner Zhou, Scott F. Grey, Felipe A. Lisboa, Desiree Unselt, Shalini Mabery, Meenu M. Upadhyay, Crystal J. Jaing, Eric A. Elster & Nicholas A. Be

Oxandrolone Efficacy in Wound Healing in Burned and Decubitus Ulcer Patients: A Systematic Review
Ana Paula C. Jalkh, Aziza K. Eastmond, Chaitra Shetty, Syed Muhammad Hannan Ali Rizvi, Joudi Sharaf, Kerry-Ann D. Williams, Maha Tariq, Maitri V. Acharekar, Sara Elena Guerrero Saldivia, Sumedha N. Unnikrishnan, Yeny Y. Chavarria, Adebisi O. Akindele, Pousette Hamid



DRYSEE LAUNCHES PATENTED WATERPROOF BANDAGE
If liquids make their way into the bandage, the gauze along its perimeter will turn a dark blue color to indicate a new bandage is needed.

Study identifies potential targets for treating venous ulcers
A comprehensive atlas of microRNA and messenger RNA expression in healing and non-healing human wounds highlights possible new approaches for treating venous ulcers – a common type of chronic, non-healing wound.


New Insights on the Effects of Dietary Omega-3 Fatty Acids on Impaired Skin Healing in Diabetes and Chronic Venous Leg Ulcers
Simona Serini and Gabriella Calviello
MolecuLight Secures Financing from BDC Canada and iGan Partners to Support its Commercial Expansion
New Financing to Meet Significant Growth in Global Demand for MolecuLight’s i:X® and DX™ Point-of-Care Imaging Devices for the Wound Care Industry
TORONTO, Aug. 11, 2022 /PRNewswire/ – MolecuLight Inc., the leader in point-of-care fluorescence imaging for real-time detection of wounds containing elevated bacterial loads, announced that it has completed a financing with BDC Capital and iGan Ventures. The funds are to support MolecuLight’s continued global expansion to meet growing customer demand for its MolecuLight i:X® and DX™ devices. Leonard Kofman and Jody Staggs, Managing Director of SWK Holdings will join MolecuLight’s Board of Directors as observers.

MolecuLight receives financing from iGan Partners and BDC Ventures (CNW Group/MolecuLight)
“With the continued growth in global demand for our i:X and DX platforms, we are happy to announce this financing from BDC Capital and iGan Ventures, who has been an early investor in MolecuLight,” says Anil Amlani, CEO of MolecuLight Inc. “The proceeds will support the continued growth of our commercial operations and infrastructure to meet market demand”.
“We have invested in MolecuLight since inception and are thrilled to see the company achieve global commercial success,” says Sam Ifergan, Founder and President of iGan Partners. “Their customers continue to generate a wealth of published data showing the improved outcomes and cost savings, which is supporting MolecuLight becoming the standard-of-care in wound care globally”.
“BDC is proud to participate in the financing of MolecuLight, an impressive Canadian company that is making a global impact in terms of improving healthcare outcomes,” says Leonard Kofman, Partner with BDC Capital’s Intellectual Property-Backed Financing practice. “MolecuLight solved an unmet clinical need – the need to detect bacterial burden in wounds, and has commercialized a suite of products that is positively impacting wound care globally. Demand for the technology is strong and growing and we believe the company is well positioned for continued growth and success”.
The MolecuLight devices are sold in North America through its direct sales and clinical applications team and internationally through MolecuLight’s 15 specialized distributors in 18 countries.
About MolecuLight Inc.
MolecuLight Inc. is a privately-owned medical imaging company that has developed and is commercializing its proprietary fluorescent imaging platform technology in multiple clinical markets. MolecuLight’s suite of commercial devices, which include the MolecuLight i:X® and DX™ fluorescence imaging systems and their accessories, are point-of-care handheld imaging devices for the real-time detection and localization of bacterial load in wounds and digital wound measurement. MolecuLight procedures performed in the United States benefit from an available reimbursement pathway which include two CPT® codes for physician work to perform “fluorescence imaging for bacterial presence, location, and load” and facility payment for Hospital Outpatient Department (HOPD) and Ambulatory Surgical Center (ASC) settings through an Ambulatory Payment Classification (APC) assignment. The company is also commercializing its unique fluorescence imaging platform technology for other global markets with relevant unmet needs in food safety, consumer cosmetics and other key industrial markets.
Tungsten Advisors served as the exclusive financial advisor to MolecuLight Inc.
About BDC Capital
BDC Capital is the investment arm of BDC, Canada’s bank for entrepreneurs. With over $3 billion under management, BDC Capital serves as a strategic partner to the country’s most innovative firms. It offers a full spectrum of capital, from seed investments to transition capital, supporting Canadian entrepreneurs who wish to scale their businesses into global champions. Visit bdc.ca/capital.
About iGan Partners:
Based in Toronto, iGan Partners is Canada’s leading health technology investors focused on disrupting the sector through breakthrough innovation that dramatically improve patent are while reducing costs. The firm focuses on identifying at an early-stage, then commercializing and scaling technological advancements in AI/Cloud-enhanced medical devices and digital health. iGan provides portfolio companies with smart-capital, active support, and access to a network of industry partners and sector-specific co-investors to help them grow and succeed.
About Tungsten Advisors:
Tungsten Advisors (www.tungstenadv.com) is an investment banking firm focused on strategic advisory and corporate finance for healthcare and technology companies. Tungsten provides transactional services including financings (private placements/PIPEs), corporate licensing and mergers and acquisitions (M&A). Tungsten also focuses on company incubation and makes direct investments alongside the creation of new companies in healthcare and technology.
Securities offered through Finalis Securities LLC Member FINRA/SIPC. Tungsten Partners LLC d/b/a Tungsten Advisors and Finalis Securities LLC are separate, unaffiliated entities.
SOURCE MolecuLight

Anemia Prevalence Among Patients With Diabetic Foot Ulcers Necessitating Surgery on Admission
A Preliminary, Retrospective Comparative Study Kaissar Yammine, Sandra Akiki, Chahine Assi, Fady Hayek

Under Pressure! Insights on Treating and Preventing Pressure Injuries
Catherine T. Milne, MSN, APRN, CWOCN-AP Amanda Estapa, ACNP-BC, CSW, FACCWS

Efficacy of Placental and Umbilical Tissue in an Infected Diabetic Foot Ulcer
Harry Schneider, DPM, FACFAS

Reflections on the Extraordinary Life of Norma N. Gill-Thompson, ET
Diane L. Krasner, PhD, RN, FAAN, FAAWC, MAPWCA
REGENATIVE LABS RELEASES NEW STUDY TO REVOLUTIONIZE TREATMENT FOR PATIENTS WITH DIABETIC FOOT ULCERS
Future applications for the early, preventative use of amniotic membrane allografts in addition to the current standard of care for DFUs present a novel opportunity to reduce long-term morbidity and amputation risk in diabetic patients.
PENSACOLA, Fla., Aug. 8, 2022 /PRNewswire/ — A new MDPI study, co-authored by Regenative Labs signifies a huge win for patients suffering from diabetic foot ulcers (DFUs). DFUs are debilitating for an individual. They are painful, long lasting, and, even with proper care, can lead to amputation. The current standard of care for DFUs is debridement, the medical removal of dead, damaged, or infected tissue to improve the healing potential of the remaining healthy tissue. The increased healing time and augmented risk of amputation associated with the current standard of care only solidifies the need for new DFU treatment alternatives. Regenerative medicine is such an alternative.
With approximately 37.3 million diabetic adults in the United States, physicians are faced with an epidemic. Given the projected increase of 1.4 million new diagnoses of diabetes mellitus each year, advancing knowledge and care for the disease and its related conditions is especially relevant.
Regenative Labs’ AmnioText™, a dehydrated amniotic membrane allograft, was used to close a grade 5 wound according to the Curative Health Services (CHS wound grade scale), described as full thickness and subcutaneous tissues, exposed tendons, ligaments, and/or joints, plus necrotic tissue in the wound, in 7 weeks. Grade 5 wounds have a 91.5% rate of not healing at all. This unsettling outcome emphasizes the efficacy and importance of amniotic membrane allografts in revolutionizing the standard of care for DFUs.
The inability to heal DFUs presents a severe danger to patients as anywhere from 5 to 24% of untreated DFUs can lead to limb amputation within 6–18 months. These infections can lead to long-term impairment and possible lower-limb amputation without timely and correct management.
“Regenative Labs and our products are focused on ameliorating this problem with our outcomes-based approach. We provide the highest quality amniotic membrane allograft to allow doctors to provide predictable outcomes for their patients,” shared Regenative Labs CEO, Tyler Barrett.
Amniotic membrane allografts, such as those provided by Regenative Labs, have proven to augment the body’s ability to regenerate the structural tissue defects associated with DFUs; they are also comparable in cost to the standard of care, which averages about USD 17,245. Not only is the current standard of care for DFUs less effective, but it is also high in cost and typically relies on inpatient expenditures.
Medicare now recognizes the medical necessity of amniotic membrane allografts in the treatment of both DFUs and venous stasis ulcers. Consequently, many patients rely on Medicare to assist with the costs associated with DFU treatment. This presents the opportunity for human amniotic membrane allografts to be utilized in rural and underserved communities where DFU treatment is typically delayed due to high costs and a lack of supplies associated with traditional treatment. This could exponentially decrease the risk of amputations in diabetic patients in these rural and underserved communities.
About Regenative Labs: Regenative Labs produces regenerative medicine products to address the root cause of a patient’s conditions using Wharton’s Jelly innovations rather than masking the pain with other treatments. Regenative Labs works closely with scientists, physicians, hospitals, and surgery centers to constantly monitor and improve patient progress and outcomes for new product development. Formed by veteran industry professionals familiar with daily challenges of innovations in healthcare, the company provides effective, non-addictive, non-invasive options for patients. Regenative Labs has a laser-focused, expert product research and development team which follows FDA guidelines of minimal manipulation for homologous use. The company adheres to AATB and FDA guidelines.
Learn more at Regenative’s website: www.regenativelabs.com
SOURCE Regenative Labs


Safety and Effectiveness of Moxibustion and Abdominal Massage in Hospitalized Older Patients With Constipation
A Retrospective Study from Kexin Li, MD


Industry education sessions brought to you by the Society of Tissue Viability
Pressure Ulcers Educational sessions specifically for people working in industry who want to refresh/upgrade their knowledge of skin health and wound management


Tackling Challenges in Wound Care by Embedding Wound Hygiene into a Proactive Wound Healing Strategy
Rachel Torkington-Stokes, MSc, BSc(Hons)


History, Current Practice, and the Future of Wound Care for Occupational and Physical Therapists
Garber, Susan L. MA, OTR, FAOTA, FACRM; O’Sullivan-Drombolis, Deirdre BScPT, MClSc (Wound Healing)

Fundamentals of pressure ulcer care
Our educational sessions promote the best practices in skin health and wound healing



Transforming Cleansing Practice | Webinar
Following the Science and Consensus Guidelines

Complex Necrotizing Fasciitis of the Abdomen and Genitals
David D Zabel, MD, FACS


Secondary Healing of Horseshoe Perianal Abscess in a Patient With Morbid Obesity
Experience at a Rural Hospital

Wound Care’s Best Kept Secret? An Easy-to-Use ECM Is Accessible to All
Wound Professionals for Use From Day 1

Etiology of Chronic Leg Ulcers in a Dermatologic Wound Clinic
A Retrospective Observational Study


Wild on Wounds National Conference | Hollywood, FL
September 7 – September 10

Chronic Non-Healing Ulcers Associated with Atopic Inflammation: A Case Report
Xianjie Yang, Huan Wang, Zhiqiang Song, Qiquan Chen Department of Dermatology, Southwest Hospital, Army Medical University, Chongqing, People’s Republic of China

Predicting Amputation in Patients With Diabetic Foot Ulcers: A Systematic Review
Zahraa Mansoor, Ali Modaweb


Rapid Detection of Pathogens in Wound Exudate via Nucleic Acid Lateral Flow Immunoassay
Anna Brunauer, René D. Verboket, Daniel M. Kainz, Felix von Stetten, and Susanna M. Früh
REGENATIVE LABS AND AAPC ANNOUNCE COLLABORATION TO FURTHER HEALTHCARE COMPLIANCE
PENSACOLA, Fla., Aug. 1, 2022 /PRNewswire/ — Regenative Labs, a leading HCT/P manufacturer, and AAPC, the nation’s largest medical coding, training, and certification association, are pleased to announce a strategic healthcare compliance collaboration. By combining AAPC’s medical training and credentialing expertise with Regenative Labs’ human tissue allografts, together both organizations will support the pioneering of the first Wharton’s jelly allografts to be assigned a Q code and be approved for application directly to a defect using a syringe.
“This is an important step in furthering the mission of educating providers and elevating healthcare outcomes and we’re proud to be a part of it,” shared AAPC CEO, Bevan Erickson.
Regenative Labs received approval from the Centers for Medicare & Medicaid Services (CMS) to cover CoreText™ and ProText™, the first Wharton’s jelly allograft products recognized as a 361 HCT/P by CMS regulated under 21CFR 1271.10, establishing a new Level II HCPCS code Q4246 “CoreText or ProText, per cc.” According to CMS, both solutions provide the extracellular matrix needed for the infiltration, attachment, and proliferation of cells required to repair damaged tissue. They are typically used for muscle and cartilage tears and help repair damaged tissue due to wounds and tissue defects and are applied directly to the defect using a syringe.
Regenative Labs, now seeking to collaborate to further HCT/P compliance, recognizes the value AAPC brings to the healthcare industry as the world’s leading healthcare association, with more than 200,00 members and 30+ years of supporting healthcare professionals, providers, payers, and health systems
AAPC will deliver healthcare providers important coding resources, including full skeletal illustrations and applicable DX codes for Regenative Labs Wharton’s jelly allografts products for specialties, including Orthopedics, Pain Management, Podiatry, and Rheumatology. In addition, AAPC will create a customized web-based training module to ensure providers understand how to utilize more specific homologous applications with current DX codes to support accurate procedural reimbursement for these products.
“We look forward to the clarity this collaboration will bring to the market allowing proper documentation of homologous use applications,” said Regenative Labs CEO Tyler Barrett.
Regenative Labs firmly believes that no other organization understands coding regulations and documentation requirements better than AAPC, leading AAPC to be the right partner for this important project.
About AAPC: AAPC’s mission is to advance the business of healthcare by providing professional training, industry-standard certifications, and comprehensive solutions to individuals and organizations across medical coding, billing, auditing, compliance, and practice management. As the most trusted source for driving accuracy, profitability, and peace of mind, AAPC helps healthcare organizations reach the full potential of their revenue cycle. Learn more at AAPC’s website: www.aapc.com
About Regenative Labs: Regenative Labs produces regenerative medicine products to address the root cause of a patient’s conditions using Wharton’s Jelly innovations rather than masking the pain with other treatments. Regenative Labs works closely with scientists, physicians, hospitals, and surgery centers to constantly monitor and improve patient progress and outcomes for new product development. Formed by veteran industry professionals familiar with daily challenges of innovations in healthcare, the company provides effective, non-addictive, non-invasive options for patients. Regenative Labs has a laser-focused, expert product research and development team which follows FDA guidelines of minimal manipulation for homologous use. The company adheres to AATB and FDA guidelines. Learn more at Regenative’s website: www.regenativelabs.com
SOURCE Regenative Labs
This article was originally published here
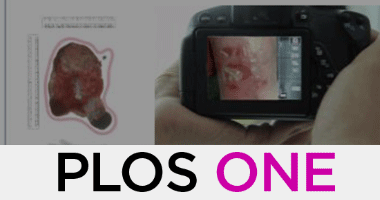
A time motion study of manual versus artificial intelligence methods for wound assessment
Heba Tallah Mohammed, Robert L. Bartlett, Deborah Babb, Robert D. J. Fraser, David Mannion


The Twin Toll of Peripheral Artery Disease and Diabetes on Minority Communities
by Elizabeth Hogan Hamacher, MHL, BSN, RN, VP of Clinical Services and Support,


Topical Biological Agents as Adjuncts to Improve Wound Healing in Chronic Diabetic Wounds
A Systematic Review of Clinical Evidence and Future Directions
The Challenges of Wound Care… “Let’s Talk Health”
Learn about wound care and healing wounds with hyperbaric oxygen therapy.
Hosted by Brian Finestein, Chief Executive Officer of Saint Clare’s Health, ‘Let’s Talk Health’ is a FREE ONLINE community series which provides accurate, trusted, and current information on health issues.





10-Year Weight Gain Substantial for US Adults
More than one-third of U.S. adults gained 10 percent body weight or more over the last 10 years, according to a study recently published in the Journal of Obesity

2 Diet Sodas A Day Could Double Risk of Diabetes
A Swedish study has found two sodas a day could double the risk of diabetes – even if they are diet versions.

Wound Healing: The Tumor Protein p53 Is at the Center of Epithelialization
The Tumor suppressing protein P53, already known as the guardian of the genome for its cancer-fighting action by which it blocks cell division in case of malignancy, is now also getting attention for its role in tissue repair in a study by molecular biologists from the universities of Bristol and Cambridge

A New Strategy for the Treatment of Chronic Wounds
Wound healing is highly regulated, but oxidative stress (OS) can disturb this healing process in chronic wounds.