BOULDER, CO / ACCESSWIRE / June 8, 2023 / Sonoma Pharmaceuticals, Inc. (Nasdaq:SNOA), a global healthcare leader developing and producing patented Microcyn® technology based stabilized hypochlorous acid (HOCl) products for a wide range of applications, including wound care, dermatology, and eye, oral and nasal care, today announced a new application for intraoperative pulse lavage irrigation treatment, which can replace commonly used IV bags in a variety of surgical procedures.
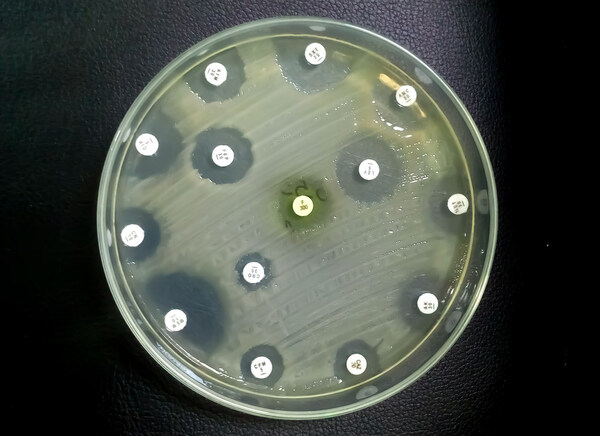
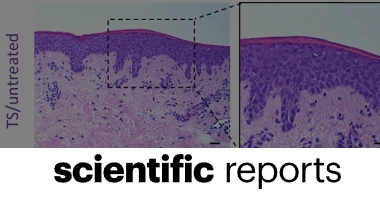
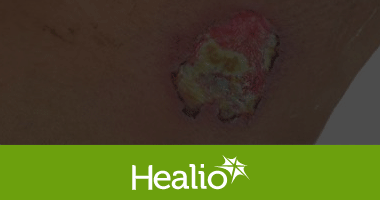
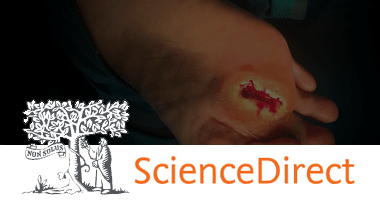
Sonoma developed this new application of its wound care technology in response to an unmet need for a non-toxic irrigation solution that can prevent infection and improve healing time. The intraoperative pulse lavage container is designed to be used in combination with a pulse lavage irrigation device, or flush gun, for abdominal, laparoscopic, orthopedic, and periprosthetic procedures. This product replaces commonly used non-antimicrobial saline and aggressive rinsing solutions with safe and effective Microcyn® Technology. Microcyn® Technology assists in the reduction of microorganisms, is non-toxic, and has regenerative properties, making it critical in preventing infection and promoting wound healing. Sonoma’s pulse lavage container is also cost competitive with IV bags, the current standard of care.
Sonoma developed the intraoperative pulse lavage irrigation treatment in close collaboration with the medical community and Sonoma’s existing distribution partners in Europe and expects this new application will be met with wide acceptance. Sonoma is now accepting orders for the pulse lavage irrigation treatment solution, which is expected to be ready for commercial use in Europe in September 2023. Sonoma anticipates commercial launch in the U.S. in 2024.
“Sonoma continues to lead in the innovation of products that improve outcomes for people with wounds or injuries or who are needing surgery. We continue to see increased demand for our wound care products in Europe, and we are excited to expand our offerings to include this next generation irrigation solution to help people heal faster following surgery,” said Amy Trombly, CEO of Sonoma Pharmaceuticals.
For more information, or to pre-order our pulse lavage irrigation treatment solution in Europe, please contact info.europe@sonomapharma.com.
About Sonoma Pharmaceuticals, Inc.
Sonoma Pharmaceuticals is a global healthcare leader for developing and producing stabilized hypochlorous acid (HOCl) products for a wide range of applications, including wound care, eye care, nasal care, oral care, dermatological conditions, animal health care and non-toxic disinfectants. The company’s products reduce infections, itch, pain, scarring and harmful inflammatory responses in a safe and effective manner. In-vitro and clinical studies of hypochlorous acid (HOCl) show it to have impressive antipruritic, antimicrobial, antiviral and anti-inflammatory properties. Sonoma’s stabilized HOCl immediately relieves itch and pain, kills pathogens and breaks down biofilm, does not sting or irritate skin, and oxygenates the cells in the area treated, assisting the body in its natural healing process. The company’s products are sold either directly or via partners in 55 countries worldwide and the company actively seeks new distribution partners. The company’s principal office is in Boulder, Colorado, with manufacturing operations in Guadalajara, Mexico. European marketing and sales are headquartered in Roermond, Netherlands. More information can be found at www.sonomapharma.com. For partnership opportunities, please contact busdev@sonomapharma.com.
Forward-Looking Statements
Except for historical information herein, matters set forth in this press release are forward-looking within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including statements about the commercial and technology progress and future financial performance of Sonoma Pharmaceuticals, Inc. and its subsidiaries (the “company”). These forward-looking statements are identified by the use of words such as “continue,” “develop,” “anticipate,” “expect” and “expand,” among others. Forward-looking statements in this press release are subject to certain risks and uncertainties inherent in the company’s business that could cause actual results to vary, including such risks that regulatory clinical and guideline developments may change, scientific data may not be sufficient to meet regulatory standards or receipt of required regulatory clearances or approvals, clinical results may not be replicated in actual patient settings, protection offered by the company’s patents and patent applications may be challenged, invalidated or circumvented by its competitors, the available market for the company’s products will not be as large as expected, the company’s products will not be able to penetrate one or more targeted markets, revenues will not be sufficient to meet the company’s cash needs, fund further development, as well as uncertainties relative to the COVID-19 pandemic and economic development, varying product formulations and a multitude of diverse regulatory and marketing requirements in different countries and municipalities, and other risks detailed from time to time in the company’s filings with the Securities and Exchange Commission. The company disclaims any obligation to update these forward-looking statements, except as required by law.
Sonoma Pharmaceuticals™ and Microcyn® are trademarks or registered trademarks of Sonoma Pharmaceuticals, Inc. All other trademarks and service marks are the property of their respective owners.
Media and Investor Contact:
Sonoma Pharmaceuticals, Inc.
ir@sonomapharmaceuticals.com
SOURCE: Sonoma Pharmaceuticals, Inc.